The compressibility factor for a real gas at high pressure is (a) 1+RT/pb (b) 1 (c) 1+pb/RT (d) 1-pb/RT - Sarthaks eConnect
$ 21.00 · 4.9 (405) · In stock
The compressibility factor for a real gas at high pressure is (a) 1+RT/pb (b) 1 (c) 1+pb/RT (d) 1-pb/RT

States Of Matter Notes: Class 11, JEE, NEET, AIIMS

SOLVED: The compressibility factor for a real gas at high pressure

Gas Compressibility - an overview

The compressibility factor of a gas is defined as Z=PV/nRT. The
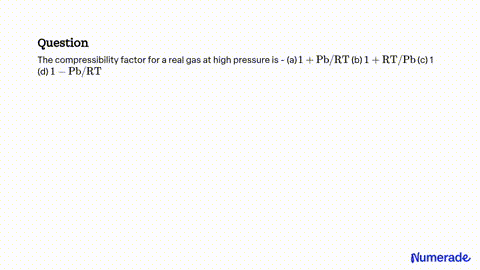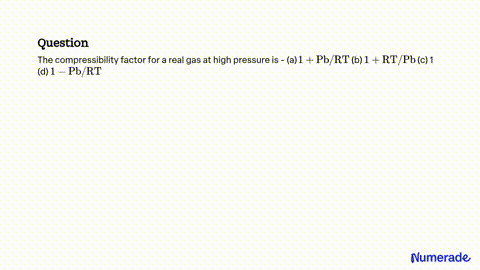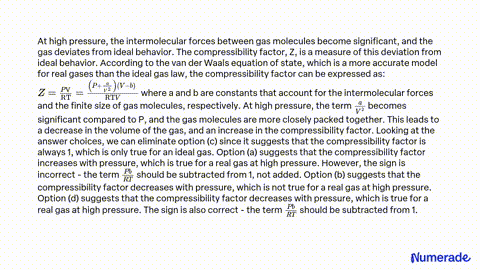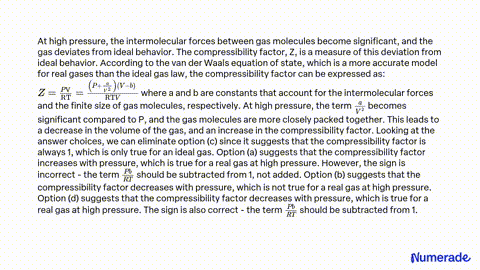
SOLVED: The compressibility factor for a real gas at high pressure
The compressibility factor for a real gas at high pressure is

The compressibility factor `(Z=PV//nRT)` for `N_(2)` at `223 K

3.2 Real gas and compressibility factor – Introduction to

Simple Equation Real Gas Compressibility Factor Z

The compressibility factor Z a low-pressure range of all gases

Compressibility Factor of Gas Overview, Equation & Chart

Calculate the compressibility factor for a gas, if 1 mole of it

States Of Matter Notes: Class 11, JEE, NEET, AIIMS

States Of Matter Notes: Class 11, JEE, NEET, AIIMS

Determine Compressibility of Gases